Ethane belongs to the Alkane series and it is a saturated hydrocarbon. Each carbon atom is connected with other atoms through a single bondYou will learn the electron dot structure of ethane.
We will discuss the electron dot structure of ethane through the following points
1.Formula of ethane
2.Electronic
configuration of carbon
3.Electronic
configuration of hydrogen
4.Find the
central atom in the compound
5.Draw the
structural formula of ethane
6.Electron
dot structure of ethane
Formula of ethane
`C_2H_6`
Electronic configuration of carbon
`1s^2 2s^2 2p^2`
Carbon has four valence electrons in the outermost
shell.
Electronic configuration of hydrogen
`1s^1`
Hydrogen has one valence electron in the outermost
shell.
Find the central atom in the compound
There are two carbon atoms and they are less in
numbers so both carbon are the central
atoms of the compound.
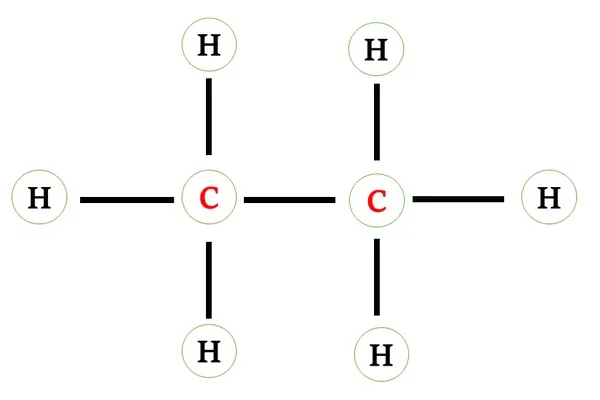
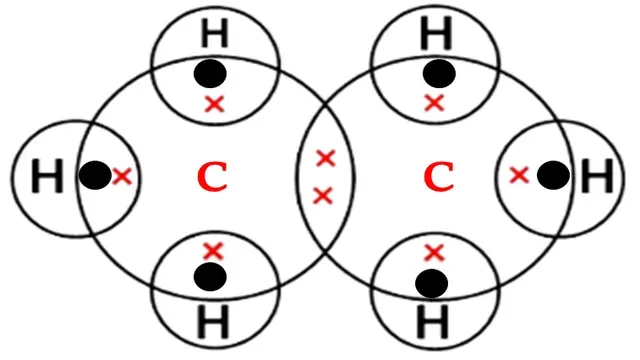
Structural formula of ethane
Carbon atom requires four electrons to complete its octet
and each hydrogen atom requires one electron to complete its octet.
Each carbon shares its three valence electrons with three
hydrogen atoms and one valence electron with another carbon atom.
A single bond (covalent bond) is present between carbon
and hydrogen atoms.




No comments:
Post a Comment