Butyne is an unsaturated hydrocarbon and belongs to the Alkyne series. in this article, we will discuss the electron dot structure of butyne.
We will discuss the electron dot structure of butyne
through the following points
1. Formula of
butyne
2. Electronic
configuration of carbon
3. Electronic
configuration of hydrogen
4. Find the
central atom in the compound
5. Draw the
structural formula of butyne
6. Electron dot
structure of butyne
Formula of butyne
`C_4H_6`
Electronic configuration of carbon
`1s^2 2s^2 2p^2`
Carbon has four valence electrons in the outermost
shell.
Electronic configuration of hydrogen
`1s^1`
Hydrogen has one valence electron in the outermost
shell.
Find the central atom in the compound
Butyne is an unsaturated hydrocarbon so carbon atoms form
a main chain.
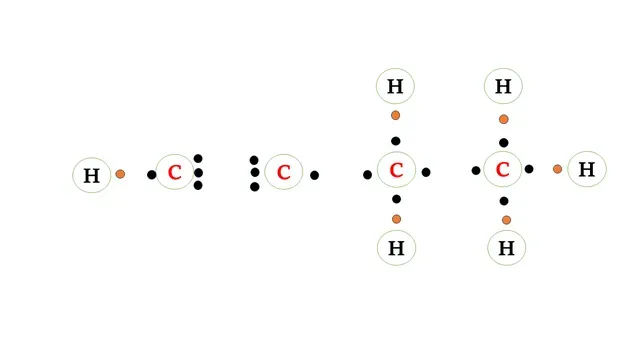
Structural formula of butyne
The carbon atom requires four electrons to complete its
octet and the hydrogen atom requires one more electron to attain a stable noble gas
configuration.
There is a triple bond between any two
carbon atoms.
Two carbon atoms share 3 -3 electrons forming a triple
bond.
The other bonds present between carbon atoms and
carbon and hydrogen atoms are single bonds that are formed after sharing 1-1
electrons.
Electron dot structure of butyne
Related Topics



.jpg)

No comments:
Post a Comment