Here, we will learn how to draw the electron dot structure of benzene. It is an organic compound that has a specific odour.
The electron dot structure of benzene will be discussed through
the following points so that you will easily understand the steps of writing the electron dot structure of benzene.
1. Formula of Benzene
2. Electronic
configuration of carbon
3. Electronic
configuration of hydrogen
4. Find the
central atom in the compound
5. Draw the
structural formula of Benzene
6. Electron
dot structure of
Formula of Benzene
`C_6H_6`
Electronic configuration of carbon
`1s^2 2s^2 2p^2`
Carbon has four valence electrons in the outermost shell.
Electronic configuration of hydrogen
`1s^1`
Hydrogen has one valence electron in the outermost
shell.
Find the central atom in the compound
Benzene is a cyclic hydrocarbon so there is no need to
find the central atom.
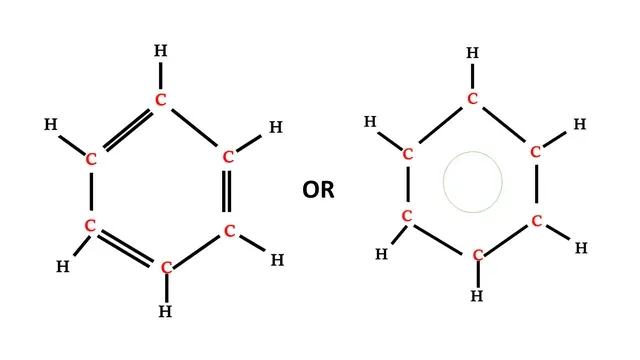
Structural formula of Benzene
Carbon atom requires four electrons to complete its octet
and each hydrogen atom requires one electron to complete its octet.
There are six carbon atoms and six hydrogen atoms.
Benzene has a hexagonal structure that includes 3 double bonds.
Double bonds are not stable between carbon atoms, they change their position frequently.
Each carbon atom is connected with one hydrogen and two other carbon atoms.
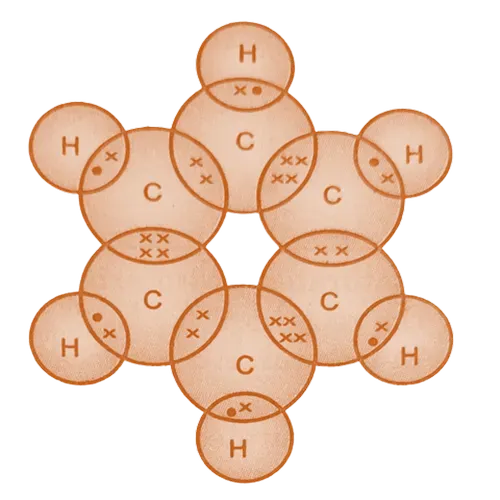
Electron dot structure of Benzene




No comments:
Post a Comment